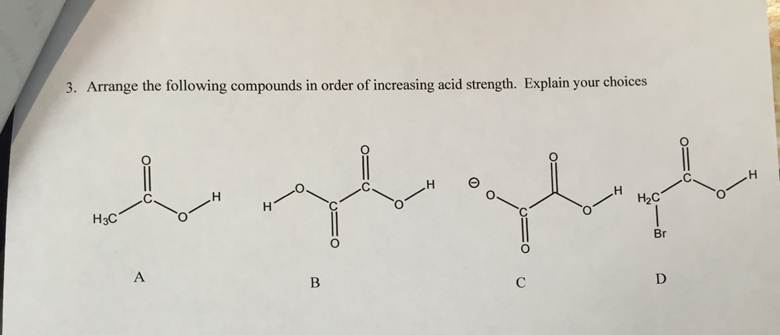
Electron withdrawing group withdraws the electrons, as a result, the electron density of phenol decreases. Arrange the compounds in order of increasing. Arrange the following compounds in the increasing order of their acidic strength: p-cresol, p-nitrophenol, phenol Hard Solution Verified by Toppr Electron-withdrawing group ( NO 2) increases the acidic character of phenols. This is because the s-character increases in the given order as the s-character increases, the electron-attracting ability increases, and the compound becomes more electronegative. Arrange the compounds in order of increasing acidity (lowest first). The two compounds containing a highly electronegative atom, i.e., the oxygen atom in their functional group, are more acidic than those with no functional. As it is well known that the order of increasing electronegativity is as sp 3 < sp 2 < sp. The given compounds will be compared based on their hybridization. The least acidic compound (second from the right) has no phenol group at. Draw the bond-line structures and arrange the following compounds in order of increasing acidity: 4-nitrobenzoic acid 4- (methylamino)benzoic acid p-chlorobenzoic acid 4- (dimethylamino)benzoic acid. Hence, the order of increasing acidity is: The order of acidity, going from left to right (with 1 being most acidic), is 2-1-4-3. 
One of the two compounds containing the –OH group includes an electron-withdrawing atom, i.e., chlorine, further increasing its acidity. The two compounds containing a highly electronegative atom, i.e., the oxygen atom in their functional group, are more acidic than those with no functional groups at all. Hence, the order of increasing acidity is: Among the groups containing –Cl and –SH, acidity increases across a row. So, the compounds containing –Cl and –SH groups are more acidic. 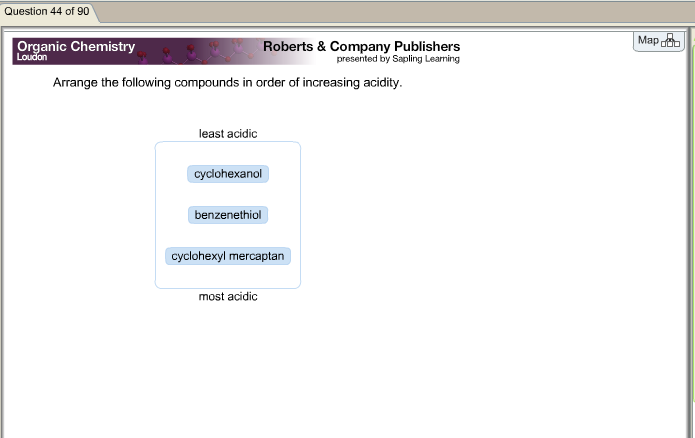
As is stated above, the presence of an electron-withdrawing group increases the acidity of the compound. Two of the three given compounds have highly electronegative atoms, acting as electron-withdrawing groups.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
